Glycerol monostearate

Stearic acid
February 6, 2018
Preservative & Parabens
February 6, 2018Competitive advantages
- Direct purchase from the manufacturer
- Fast delivery of the product
- Providing the best brands in the world
Monostear glycerol; application, properties and properties
Glycerol monostearate (Glycerol Monostearate) with the chemical formula C21H24O4 is used as a raw material for the production of various edible and industrial products. This material is in fact a stabilizer or stabilizer and is derived from laboratory synthesis of substances such as glycerin and fatty acids. It is naturally present in soybean oil. Cakes, Chocolate, Butter Creams are the most important food products that use monosodium glycerol as an additive.
Our brands
- Irmal - Malaysia
Physical, chemical and other properties of monostear glycerol:
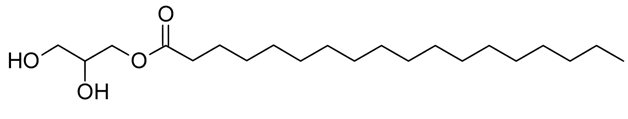
Glycerol is a molecular mono-stearate made of oxygen, hydrogen, and carbon atoms.
This chemical is known as the GMS abbreviation in the market. In the structure of this molecule, there is a group of ester and hydroxy. The molecular structure of glycerol mono-stearates is shown in the figure below.
Glycerol monostearates are in two forms of isomers. The name of IUPAC is the chemical, 2 and 3-dihydroxypropyl octo-decanoate. The physical and chemical characteristics of monostear glycerol are shown in the table below.
| Physical and chemical properties | Description |
|---|---|
| physical state | Solid white |
| Molecular formula | C21H42O4 |
| Molecular weight | 56/358 |
| Common names | 2 and 3-dihydroxypropyl octo-decanoate / glyceryl mono-stearate / glycerin mono-stearate / mono-stearin |
| Isomers | 1-monostearate glycerol 2-monostearate glycerol |
| Melting point (° C) | 68 to 63 |
| viscosity | 180 تا 220 |
| Density (grams per cubic centimeter) | 03/1 |
| Boiling point (° C) | More than 100 degrees |
| Refractive index | 432/1 |
| Flash point (fahrenheit) | More than 400 degrees |
| Storage and maintenance conditions | room temperature |
| Solubility in water | Insoluble |
| Solubility in other solvents | Particular solubility in ethanol / insoluble in aliphatic solvents |
Applications of glycerol monostearate:
Monostearate glycerol is used as an additive to produce a variety of health and beauty products, medications and all kinds of foods. The main use of glycerol mono-stearates is its use as an emulsion. Ice cream companies, edible oils, cream and bread, butter, and jelly desserts, dairy products, and many beverages use monostearate glycerol to produce their own products. The most important uses of this substance are listed in the table below.
| Industry name | Application of monostearate glycerol |
|---|---|
| Pharmacy | Emulsifier in the production of medications |
| make up products | Cream, Powder & Skin Care / Face Makeup / Softeners / Sunscreens / Skin Moisturizer |
| Food industry | Amber / Flavor / Food Stabilizer / Candy, Chocolate, Chewing Gum / Cake Making / Preparing a variety of cooked foods / Preparing Biscuits / Preparing Various Oils & Fats / Beverages Emulsifiers / Drink Flavors |
| Industrial applications | Textile Industrial Emulsion / Industrial Oils / Film Production for Plastics / Moisture / Waterproofing |
Safety Notes and Cautions When working with chemical glycerol Mono Stearates:
Regarding the molecular structure of monostear glycerol, it is observed that active sites are involved in chemical reactions in different parts of the molecule; the carbonyl group, the stereonic group, and the hydroxy groups in the monostearate glycerol molecule are susceptible to reactions They are chemical, so people who deal with this chemical should be well aware of the complications and risks.
For monostearate glycerol, no toxic and destructive effects have been recorded, but if it is contacted by the skin or eyes, it will cause irritation and severe inflammation. Inhalation of glycerol mono-stearates will produce chronic effects on the respiratory system.
If your eyes or skin contact with glycerol mono-stearates, you should quickly wash the area with water and consult your doctor if inflammation is increased. If someone randomly swallows glycerol mono-stearate, he should immediately bring him to the treatment center.
If the temperature of the glycerol mono-stearate is increased, the flammability of the product is burned, and carbon dioxide and carbon monoxide are produced by burning. The best way to store and store this material is to store it in a place far from moisture and source of heat and heat. This material should be stored in closed containers and in a cool place.




